Crystallography
Crystallography is the experimental science of determining the arrangement of atoms in solids. In older usage, it referred to the scientific study of crystals.
Before the development of X-ray diffraction crystallography, the study of crystals was based on their geometry. It involved measuring the angles of crystal faces relative to theoretical reference axes (crystallographic axes), using a goniometer, and establishing the symmetry of the crystal in question.
Crystallographic methods now depend on the analysis of the diffraction patterns that emerge from a sample that is targeted by a beam of some type. The beam is not always electromagnetic radiation, although X-rays are the most common choice. For some purposes, electrons or neutrons are used, which is possible due to the wave properties of the particles. Crystallographers often explicitly state the type of illumination used when referring to a method, as with the terms X-ray diffraction, neutron diffraction, and electron diffraction.
Etymology
The word "crystallography" is derived from the Greek words crystallon = cold drop / frozen drop, with its meaning extending to all solids with some degree of transparency, and graphein = write.
Three types of crystallography
The three types of radiation—X-rays, electrons, and neutrons—interact with the specimen in different ways. X-rays interact with the spatial distribution of the valence electrons, while electrons are charged particles and therefore feel the total charge distribution of both the atomic nuclei and the surrounding electrons. Neutrons are scattered by the atomic nuclei through the strong nuclear forces, but in addition, the magnetic moment of neutrons is non-zero. They are therefore also scattered by magnetic fields. Because of these different forms of interaction, the three types of radiation are suitable for different crystallographic studies.
Theory
An image of a small object is usually generated by using a lens to focus the illuminating radiation, as is done with the rays of the visible spectrum in light microscopy. However, the wavelength of visible light (about 4000 to 7000 Angstroms) is three orders of magnitude longer then the length of typical atomic bonds and atoms themselves (about 1 to 2 Angstroms). Therefore, obtaining information about the spatial arrangement of atoms requires the use of radiation with shorter wavelengths, such as X-rays. Employing shorter wavelengths implied abandoning microscopy and true imaging, however, because there exists no material from which a lens capable of focusing this type of radiation can be created. (That said, scientists have had some success focusing X-rays with microscopic Fresnel zone plates made from gold, and by critical-angle reflection inside long tapered capillaries). Diffracted x-ray beams cannot be focussed to produce images, so the sample structure must be reconstructed from the diffraction pattern. Sharp features in the diffraction pattern arise from periodic, repeating structure in the sample, which are often very strong due to coherent reflection of many photons from many regularly spaced instances of similar structure, while non-periodic components of the structure result in diffuse (and usually weak) diffraction features.
Because of their highly ordered and repetitive structure, crystals give diffraction patterns of sharp Bragg reflection spots, and are ideal for analyzing the structure of solids.
Technique
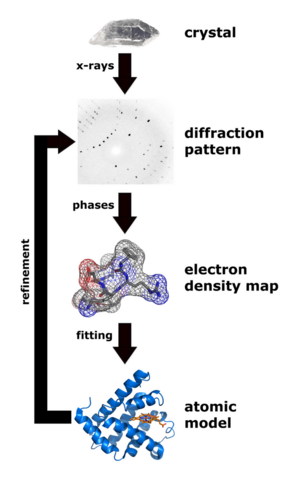
Some materials studied using crystallography, proteins for example, do not occur naturally as crystals. Typically, such molecules are placed in solution and allowed to crystallize over days, weeks, or months through vapor diffusion. A drop of solution containing the molecule, buffer, and precipitants is sealed in a container with a reservoir containing a hygroscopic solution. Water in the drop diffuses to the reservoir, slowly increasing the concentration and allowing a crystal to form. If the concentration were to rise more quickly, the molecule would simply precipitate out of solution, resulting in disorderly granules rather than an orderly and hence usable crystal.
Once a crystal is obtained, data can be collected using a beam of radiation. Although many universities that engage in crystallographic research have their own X-ray producing equipment, synchrotrons are often used as X-ray sources, because of the purer and more complete patterns such sources can generate. Synchrotron sources also have a much higher intensity of X-ray beams, so data collection takes a fraction of the time normally necessary at weaker sources.
Producing an image from a diffraction pattern requires sophisticated mathematics and often an iterative process of modeling and refinement. In this process, the mathematically predicted diffraction patterns of an hypothesized or "model" structure are compared to the actual pattern generated by the crystalline sample. Ideally, researchers make several initial guesses, which through refinement all converge on the same answer. Models are refined until their predicted patterns match to as great a degree as can be achieved without radical revision of the model. This is a painstaking process, made much easier today by computers.
The mathematical methods for the analysis of diffraction data only apply to patterns, which in turn result only when waves diffract from orderly arrays. Hence crystallography applies for the most part only to crystals, or to molecules which can be coaxed to crystallize for the sake of measurement. In spite of this, a certain amount of molecular information can be deduced from the patterns that are generated by fibers and powders, which while not as perfect as a solid crystal, may exhibit a degree of order. This level of order can be sufficient to deduce the structure of simple molecules, or to determine the coarse features of more complicated molecules (the double-helical structure of DNA, for example, was deduced from an X-ray diffraction pattern that had been generated by a fibrous sample).
Materials engineering
Crystallography is a tool that is often employed by materials scientists. In single crystals, the effects of the crystalline arrangement of atoms is often easy to see macroscopically, because the natural shapes of crystals reflect the atomic structure. In addition, physical properties are often controlled by crystalline defects. The understanding of crystal structures is an important prerequisite for understanding crystallographic defects. Mostly, materials do not occur in a single crystalline, but poly-crystalline form, such that the powder diffraction method plays a most important role in structural determination.
A number of other physical properties are linked to crystallography. For example, the minerals in clay form small, flat, platelike structures. Clay can be easily deformed because the platelike particles can slip along each other in the plane of the plates, yet remain strongly connected in the direction perpendicular to the plates. Such mechanisms can be studied by crystallographic texture measurements.
In another example, iron transforms from a body-centered cubic (bcc) structure to a face-centered cubic (fcc) structure called austenite when it is heated. The fcc structure is a close-packed structure, and the bcc structure is not, which explains why the volume of the iron decreases when this transformation occurs.
Crystallography is useful in phase identification. When performing any process on a material, it may be desired to find out what compounds and what phases are present in the material. Each phase has a characteristic arrangement of atoms. Techniques like X-ray diffraction can be used to identify which patterns are present in the material, and thus which compounds are present (note: the determination of the "phases" within a material should not be confused with the more general problem of "phase determination," which refers to the phase of waves as they diffract from planes within a crystal, and which is a necessary step in the interpretation of complicated diffraction patterns).
Crystallography covers the enumeration of the symmetry patterns which can be formed by atoms in a crystal and for this reason has a relation to group theory and geometry. See symmetry group.
Biology
X-ray crystallography is the primary method for determining the molecular conformations of biological macromolecules, particularly protein and nucleic acids such as DNA and RNA. In fact, the double-helical structure of DNA was deduced from crystallographic data. The first crystal structure of a macromolecule, myoglobin, was solved in 1958.[1]
The Protein Data Bank (PDB) is a freely accessible repository for the structures of proteins and other biological macromolecules. Computer programs like RasMol or Pymol can be used to visualize biological molecular structures.
Electron crystallography has been used to determine some protein structures, most notably membrane proteins and viral capsids.
Notations used in crystallography
- Coordinates in square brackets such as [100] denote a direction vector (in real space).
- Coordinates in angle brackets or chevrons such as <100> denote a family of directions which are related by symmetry operations. In the cubic crystal system for example, <100> would mean [100], [010], [001] or the negative of any of those directions.
- Miller indices in parentheses such as (100) denote a plane of the crystal structure, and regular repetitions of that plane with a particular spacing. In the cubic system, the normal to the (hkl) plane is the direction [hkl], but in lower-symmetry cases, the normal to (hkl) is not parallel to [hkl].
- Indices in curly brackets or braces such as {100} denote a family of planes and their normals which are equivalent due to symmetry operations, much the way angle brackets denote a family of directions.
See also
- Crystal
- Crystallization
- Diffraction
- DNA
- Electron
- Ion (physics)
- Neutron
- Optics
- Protein
- RNA
- X-ray
- X-ray crystallography
Notes
- ↑ J.C. Kendrew, et al. 1958. A three-dimensional model of the myoglobin molecule obtained by X-ray analysis. Nature 181:662–666.
ReferencesISBN links support NWE through referral fees
- Guinier, André. X-Ray Diffraction in Crystals, Imperfect Crystals, and Amorphous Bodies. reprint ed. New York: Dover, 1994 (original 1963). ISBN 0486680118
- Hammond, Christopher. The Basics of Crystallography and Diffraction, 2nd ed. Texts on Crystallography, 5. Oxford, UK: Oxford Univ. Press, 2001. ISBN 0198505523
- Kendrew, J.C. et al. A three-dimensional model of the myoglobin molecule obtained by X-ray analysis. Nature 181 (1958): 662–666.
- Mullin, J. W. Crystallization, 4th ed. Oxford: Lutterworth-Heinemann, 2001. ISBN 978-0750648332
- Sands, Donald. Introduction to Crystallography. Dover Classics of Science and Mathematics. New York: Dover, 1993. ISBN 0486678393
External links
All links retrieved January 11, 2024.
- SINCRIS Information Server for Crystallography
- Commission on Crystallographic Teaching, Pamphlets
- Crystallography site of Steffen Weber with lots of Java Applets
- IUCr Online Dictionary of Crystallography
Credits
New World Encyclopedia writers and editors rewrote and completed the Wikipedia article in accordance with New World Encyclopedia standards. This article abides by terms of the Creative Commons CC-by-sa 3.0 License (CC-by-sa), which may be used and disseminated with proper attribution. Credit is due under the terms of this license that can reference both the New World Encyclopedia contributors and the selfless volunteer contributors of the Wikimedia Foundation. To cite this article click here for a list of acceptable citing formats.The history of earlier contributions by wikipedians is accessible to researchers here:
The history of this article since it was imported to New World Encyclopedia:
Note: Some restrictions may apply to use of individual images which are separately licensed.